What sparked my interest to create this article is the questionable statement by Biologist Francis Mangel during his interview with John Whyte, which claimed that spraying is not a threat yet. You can watch it on You Tube a video with title: “Francis Mangels at Consciousness Beyond Chemtrails 2012” His statement is an answer to a question,27 minutes into the video:
Question: “(John Whyte): Question from July from southern California, he wants to know what effects from geoengineering are having on the primary roots system in crop, trees, etc.
Answer: (Francis Mangels): “Nothing yet. But hold on for that. If it gets really high we’ll see roots stunting from heavier aluminum, barium and strontium tends to replace calcium. We’ll probably see some deficiency disease, eventually, it could be global warming, that weaken the plants predisposes, for instance the western coast department deal with that, it could be global warming. But is there anything that poison the root of the plant now? not yet. The big thing is pH change, its debilitating the planet a little bit now, but in a word, no, no poison. pH changes are probably more dangerous, and global warming probably more dangerous at this time”

I disagree with the above statement. I believe that the spraying did kill plants. Maybe not by means of aluminium, but by other means like drought or acid rain. I will discuss the above statement one claim at a time. Francis Mangels seems to refer the trails problem when geoengineering mentioned, let us call it the proper way, chemtrails or the spraying. Since Francis Mangels considers aluminium as the main problem of chemtrails, this article will only discuss aluminium.
Can aluminium poison the root of the plants?
First we have to keep in mind that aluminium has many forms. Many people simply call it aluminium, but when aluminium itself bonds to different atom, it becomes different. It produces a new form with different properties, which also includes a different toxicity. The toxicity of aluminium depends greatly on its form. Below is simply one example of Aluminum Oxide.
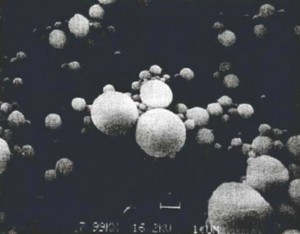
Above photo courtesy of Aerospace Web http://www.aerospaceweb.org/question/propulsion/q0246.shtml
- “Why is aluminum used in solid rocket propellants? What is the role of aluminum agglomeration in the combustion of propellants?”
“A solid rocket is a class of rocket in which the fuel and oxidizer are mixed together and cast into a solid material. This solid material is typically formed into cylindrical blocks with a void along the centerline. Once exposed to an ignition source, a flame travels through this empty central region and consumes the propellant grain radially until the fuel supply is exhausted. A liquid rocket, on the other hand, has separate fuel and oxidizer tanks in which the two substances are stored as fluids until they are combined and burned in the rocket’s combustion chamber. The two types of rockets have their advantages and disadvantages as described in a previous article on jets and rockets. Although liquid rockets are typically more powerful and efficient, solid rockets are usually less complex and safer to store. Many techniques exist to enhance the performance of solid rocket motors. Adding chunks of aluminum to the mixture of fuel and oxidizer is one common technique to increase the thrust a solid rocket produces.”
So is it important to know what kind of aluminium is being released by the jet planes. According to “What in the World Are They Spraying”, at 00:08:57: it states: “The little picture is from a nano-fabrication study, which shows you can make very high quality and do this with a jet just in a very simple way make high quality aluminum particles just by spraying aluminum vapor out which oxydizes.”
The aluminium that is currently being sprayed will oxidize.
The above statement suggests to me that the metal particle fall out will be nano aluminium oxide. Anyone have a problem with this? If you do, check the following:
According to Material safety datasheet of Dana Ridge: “Aluminium Oxide Al2O3 Not classified as hazardous according to criteria of worksafe australia” It specify the color of aluminium oxide as brown, bluish, grey, black, white, and pink. pH value as N/A. Also mention “no special action required” for inhalation, skin contact and even ingestion.
According to Material safety datasheet of Laguna Clay company. “NOT EXPECTED TO PRODUCE SIGNIFICANT ADVERSE HEALTH EFFECTS WHEN THE RECOMMENDED INSTRUCTIONS FOR USE ARE FOLLOWED.” But also: “Exposure to airborne concentrations above statutory or recommended exposure limits may cause irritation of the nose, throat and lungs. No known significant effects or critical hazards for Ingestion and Skin. Chronic effects Repeated or prolonged inhalation of dust may lead to chronic respiratory irritation.”
According to Material safety datasheet of Santa Cruz Biotechnology, Inc. “Aluminum oxide CONSIDERED A HAZARDOUS SUBSTANCE ACCORDING TO OSHA 29 CFR 1910.1200.“ Also mentions produce use as: “Used as an adsorbent, desiccant. As a filler in paints & adhesives. In the manufacture of alloys, ceramics, electrical insulators, resistors, dental cements, glass. As an abrasive and in metal polishes. In the manufacture of artificial gems, in coatings for metals. Gamma alumina – as a catalyst / catalyst support; used in chromatography.” “The material has NOT been classified as “harmful by ingestion”. This is because of the lack of corroborating animal or human evidence. The material may still be damaging to the health of the individual, following ingestion, especially where preexisting organ (e.g. liver, kidney) damage is evident. Present definitions of harmful or toxic substances are generally based on doses producing mortality (death) rather than those producing morbidity (disease, ill-health). Gastrointestinal tract discomfort may produce nausea and vomiting. In an occupational setting however, unintentional ingestion is not thought to be cause for concern.” “The material is not thought to produce respiratory irritation (as classified using animal models). Nevertheless inhalation of dusts, or fume, especially for prolonged periods, may produce respiratory discomfort and occasionally, distress. Inhalation of dusts, generated by the material during the course of normal handling, may be damaging to the health of the individual. The inhalation of small particles of metal oxide results in sudden thirst, a sweet, metallic our foul taste, throat irritation, cough, dry mucous membranes, tiredness and general unwellness. Headache, nausea and vomiting, fever or chills, restlessness, sweating, diarrhea, excessive urination and prostration may also occur. After exposure is removed, recovery occurs within 24-36 hours.”
“Limited evidence suggests that repeated or long-term occupational exposure may produce cumulative health effects involving organs or biochemical systems. Chronic exposure to aluminas (aluminium oxides) of particle size 1.2 microns did not produce significant systemic or respiratory system effects in workers.” “There is general agreement that particle size determines that the degree of pathogenicity (the ability of a micro-organism to produce infectious disease) of elementary aluminium, or its oxides or hydroxides when they occur as dusts, fumes or vapours. Only those particles small enough to enter the alveolii (sub 5 um) are able to produce pathogenic effects in the lungs. Exposure to large doses of Aluminum has been connected with the degenerative brain disease Alzheimer’s Disease.”

According to “What in the World Are They Spraying”, at 00:31:55, statement of Dr. Lenny Thyme, PhD in inorganic chemistry from Oregon State University stated:
“So, once it gets to the aluminum oxide stage it just forms a plaque within your arteries and SHUTS DOWN LIFE! And so, as you accumulate aluminum over time, it causes major neurological damage, because it ends up as aluminium oxide that´s essentially stuck in place and can´t be flushed out by normal systems.” Note that he mention aluminium as an element.”
Previous statements from the same video indicates that the released aluminium should fall as aluminium oxide, not as aluminium element.
Confused yet?
Is aluminium oxide really safe for the environment?
Most of the above statements do suggest that aluminium oxide is relatively safe. But is it really safe for the plants?
But to add to the confusion, the same document claims that Aluminum really isn’t safe after all.
“Aluminium occurs in the environment in the form of silicates, oxides and hydroxides, combined with other elements such as sodium, fluorine and arsenic complexes with organic matter. Acidification of soils releases aluminium as a transportable solution. Mobilization of aluminium by acid rain results in aluminium becoming available for plant uptake.” Acid or alkaline environment will make aluminium oxide harmful. “The bioavailability and toxicity of aluminium is generally greatest in acid solutions. Aluminium in acid habitats has been observed to be toxic to fish and phytoplankton . Aluminium is generally more toxic over the pH range 4.4.5.4, with a maximum toxicity occurring around pH 5.0.5.2 . The inorganic single unit aluminium species (Al(OH)2 +) is thought to be the most toxic. Under very acid conditions, the toxic effects of the high H+ concentration appear to be more important than the effects of low concentrations of aluminium; at approximately neutral pH values, the toxicity of aluminium is greatly reduced. The solubility of aluminium is also enhanced under alkaline conditions, due to its amphoteric character, and some researchers found that the acute toxicity of aluminium increased from pH 7 to pH 9 . However, the opposite relationship was found in other studies. The uptake and toxicity of aluminium in freshwater organisms generally decreases with increasing water hardness under acidic,neutral and alkaline conditions. Complex agents such as fluoride, citrate and humic substances reduce the availability of aluminium to organisms, resulting in lower toxicity. Silicon can also reduce aluminium toxicity to fish.”
Another document also show that aluminium can hold back plants growth: Toxicity-Resistant Crops – Researchers have engineered aluminum-tolerant crops “Aluminum is common in soils–it’s a major component of clay–but only in acidic soils does the metal form an ion that can dissolve into liquids and that’s toxic to plants. Acidic soils make up as much as half the world’s croplands, however, and aluminum toxicity is the main factor holding back crop growth in nearly 20 percent of the world’s arable soils, including large areas of the United States east of the Mississippi River and northwestern Europe.” “Toxic aluminum ions are known to damage DNA, and the new study suggests that plants respond by shutting down growth of cells in the tips of their roots when they accumulate too much DNA damage. Plants may have evolved this response to help them, over generations, cope with aluminum’s toxic effects, Larsen speculates.
But in the short run, it means that the plants are less healthy and are stunted and more vulnerable to stressors such as droughts.” ““The work provides the first compelling evidence for a mechanism that explains the toxic effect of [aluminum] on root growth,” says plant biologist Manny Delhaize of the Commonwealth Scientific and Industrial Research Organisation Plant Industry Center, in Canberra, Australia. “There have been numerous theories about how aluminum arrests root growth, and this work provides convincing evidence regarding the molecular process involved.” Delhaize says that another method of keeping the growth rates high, while limiting any DNA damage, might be to engineer plants so that their root tips express molecules that would inactivate AtATR.”
In summary, Francis Mangels is wrong. Aluminium already able to poison the plants today.
Will aluminium oxide neutralize the soil?
According to Yahoo Answer – What is the product for the reaction of aluminum oxide and sulfuric acid?
“Al2O3 + 3H2SO4 ——– Al2(SO4)3 + 3 H2O
The compound forms a hexahydrate in aqueous systems (aqueous H2SO4).”
So yes, aluminium oxide will neutralize the soil. It will reduce the acidity of soil, and make soil more neutral.

Above photo see : http://www.350resources.org.uk/2011/12/03/spectator-magazine-says-global-warming-is-all-a-myth-by-george-monbiot/
Can chemtrails poison the plants?
Yes, definitely, even without aluminium, chemtrails is still harmful to plants, crops, animals and humans.
Please read the our article regarding sulfur and chemtrails. The spraying will reduce ozone layer, create acid rain, create drought and prevent the plants from receiving full sunlight.
Is pH changes more dangerous than the spraying?
This discussion is still open for debate.
Is global warming more dangerous than the spraying?
I do not believe in Global Warming hoax. Our planet is not in danger of melting, but your weather may already become wild because the spraying. I believe that chemtrails is the cause of global warming. The spraying is the real threat, not the global warming. The spraying is the problem, global warming is nothing.



The sky is full of aluminum oxide mixed with hydrogen turns it into rocket fuel.I have spent 50 years welding aluminum it is in my lungs,my doctor said it is my fault,i chose my profession,aircraft fuel contains 16 per cent aluminum,an aircraft burns 65 gallons a minute,10 tons per hour.25.000 aircraft in the sky 24 hours a day 7 days a week.The a380 aircraft burns 14 tons per hour.Governments are conning the public to such an extent something has got to be done.It is killing trees and plants.it causes altzeimers and demenchure (sorry about the spelling) I am a sick disabled pensioner,aluminum gets into your brain and lungs,aircraft fuel (paraffin) is the dirtiest fuel on the planet,the V.O.C. s ,(volatile organic compounds) stick to your lungs.People should stay well clear of airports.I could go on but i am struggling to breath. Thank you for you time. Tony. S.
I could only watch a third of this video because it was so disturbing. It is to real and I feel so inadequate and guilty for doing nothing to stop this from happening. If you have the mental sturdiness to watch this I applaud you. I am now trying to find like minded people to gather and find a solution something everyone can help with. Please watch this video it truly is the most important video at this time to watch since if we do nothing about this now we will never have to worry again for there will be no future to worry about. I am ashamed that I took so long to wake up but I am awake now and so are you lets get this thing done. I am not a scientist but I have lips and I can talk real long and loud I just have to have facts about what to do.
http://www.youtube.com/watch?v=ZUkkpvXR-zk&w=680&h=440
The Most Important Video of 2013! – Nsearch Radio
May I ask what part of this was most disturbing to you as well we would love to get your email address as well work with you on anything that you have the capability of handling at this time, as well thank you for your comment!
Evone, don’t let that video scare you. There is no hidden catastrophy. Chemtrails is serious problem, but we can protect ourself, our plants, our animal and our environment.
If the end result you want is better weather, please try cemenite.
Thanks, Roxi, for taking the time to research this question and share the referenced material here with us. I really appreciate your contribution to the TRUTHER MOVEMENT – it’s websites like the TRUTH DENIED that are making information available and easy to understand,. so that more and more people are finally looking up. I have been studying the sky and photographing the massive aerosol spraying operation happening here in Northern California since 2000. I was a letter carrier with the USPS and had delivered mail on a walking route near the Sacramento River for 16 years — I walked through the webs of nanoparticles and breathed in the dust of aluminum and barium that was layered on the mailboxes and it was easy to see the effect of the spray on the landscape. I would see the jets lay down the patterned trails across a clear blue sky in the morning and then watch them spread into a nasty grey haze that covered the sun by the afternoon. When I tried to talk with my coworkers, family and friends about the subject, they mostly just rolled their eyes at me, and wouldn’t even look at the facts. It was really strange and I’m sure that they thought that I was crazy. So, for the past 13 years I’ve continued to do my own silent study and I’m encouraged by the quality information on the web. People are realizing that this is not a conspiracy theory anymore and they are beginning to look up and ask questions — knowledge is power!
Thank your for a very refreshingly logical treatment of this subject. You don’t get this quality of critical thinking on the internet very often. I will do my best to harvest old links and pass them on regarding the fact that aluminum gels the inside of plant circulatory systems the same way that Alum for pickles sets up pickles and our own lymphatic system. Lymph stasis by aluminum will kill us just as sure as aluminum will kill a plant.